HRPP Overview
- HRPP -- Everything You Wanted To Know
- HRPP Toolkit Overview
- What Is An Institutional Official?
- What to Do If Things Go Wrong
The HRPP stands for the Human Research Protection Program. The Harvard University Area HRPP is a collaborative effort between all who develop, conduct, review, approve and facilitate Human Research. It includes institutional leaders, the Institutional Review Board (IRB), researchers, faculty, staff, students and the community. The HRPP aims to protect the rights and welfare of research volunteers, by providing support, guidance, and education to facilitate research that is ethical and scientifically sound.
The HRPP Plan provides an overview of the Human Research Protection Program at Harvard University. The document defines roles, our mission, and includes a complete outline of the regulatory requirements that must be adhered to. It is the "30,000 foot view" document that ties all of the pieces of our program together.
HRPP Toolkit
The Policies, Procedures, Guidance, and Documents that are part of the Human Research Protection Program at Harvard University Area IRB are collectively known as the “Toolkit”. The Toolkit consists of “living” documents that will be updated as our regulatory landscape changes. We will also provide additional guidance and IRB decision aids to assist the research community through the IRB review process. Here is an overview of the components of the Toolkit:
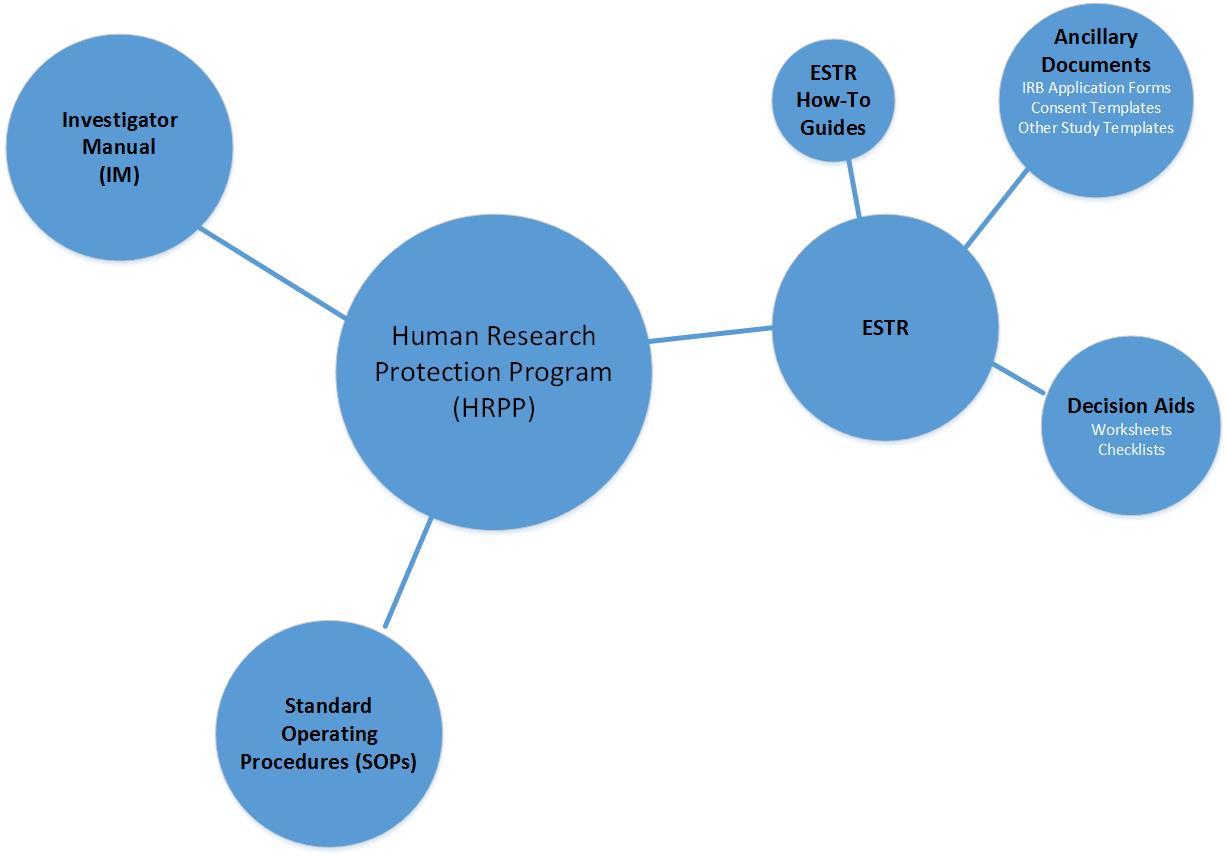
- Human Research Protection Program (HRPP) Plan: The HRPP Plan provides an overview of the Human Research Protection Program at Harvard University. The document defines roles, mission, and includes a complete outline of the regulatory requirements that must be adhered to. It is the "30,000 foot view" document that ties all of the pieces of our program together.
- Investigator Manual (IM): The IM provides additional guidance and information on various regulatory topics, insight into nebulous areas of IRB review, and resources that assist in navigating the IRB process.
- Standard Operating Procedures (SOPs): SOPs act as our “instruction manual.” They are comprised of the “nuts and bolts” of the IRB review process in our electronic submission system (ESTR) and policy that guides the human subject research review process. SOPs are found in the last tab of the ESTR Library.
- Electronic Submission, Tracking, and Reporting (ESTR): ESTR is the electronic submission system that automates the IRB review process for the Harvard IRBs (Harvard University Area and Longwood Medical Area).
ESTR How-To Guides
These guides provide step-by-step instructions for the ESTR system according to the user profile:
- Study Submission Guide: for Principal Investigators and study staff
- Ancillary Reviewer Guide: for faculty sponsor, department or other non-IRB ancillary reviewers
- Study Reviewer's Guide: for IRB reviewers
- IRB Staff Administration Guide: for IRB staff
ESTR Decision Aids
The ESTR system library (you must be logged into ESTR) contains resourceful worksheets and checklists.
- Checklists are completed by IRB staff to document regulatory decisions according to the human subject protection regulations. Worksheets can also be helpful for researchers to gain insight into the regulatory determinations that are being made.
- Worksheets are used by IRB staff and researchers to assist in regulatory decisions. IRB staff may also request that specific worksheets be completed by researchers to gather information for a particular regulatory determination.
Ancillary Documents
The ESTR system library and IRB website list the forms and templates that are needed to complete an IRB submission. Ancillary documents are available in the ESTR system and provide needed regulatory information for a study.
- IRB Submission Forms: The CUHS Protocol Template is the main application form that is used to submit all IRB submissions with the exception of a Not Human Subjects Research Determination as there is a separate form for that type of submission. Both forms have been revised to gather important information and reduce the back and forth that occurs from not having all the required information at the outset.
- Supplemental Forms: These forms include the Financial Interest Disclosure Form when there is a financial interest to disclose as well forms to complete when including non-Harvard personnel in a study or when there is a request for a reliance agreement between IRBs. The ESTR system contains questions that let researchers know when one or more of these forms is needed. If one is missed, don’t worry – IRB staff will let you know.
- Consent Form Templates: The regulations require that certain elements are included in consent forms that are provided to subjects in research. There are also specific elements required if a research study includes sharing of genetic data or if there is a Certificate of Confidentiality, to name just a few. The consent form templates include all required elements that may needed. If a certain element does not apply, then just delete that section from the template form.
The University Chief Research Compliance Officer, Ara Tahmassian, Ph.D., is designated as the Institutional Official/Organizational Official (IO/OO) for the Harvard University HRPP.
The IO/OO has the authority to:
- Create policies and procedures related to the HRPP.
- Suspend or terminate research approved by the IRB.
- Disapprove research approved by the IRB.
- And, a lot of other authority – check out the HRPP Plan for the complete listing.
The IO/OO also has many responsibilities in the HRPP, including (but not limited to):
- Oversee the review and conduct of Human Research under the jurisdiction of the HRPP.
- Periodically review the HRPP plan to assess whether it is providing the desired results and recommend amendments as needed.
- Establish policies and procedures designed to increase the likelihood that Human Research will be conducted in accordance with ethical and legal requirement.
- Ensure that the research review process is independent and free of coercion or undue influence and ensure that officials of the Institution cannot approve research that has not been approved by the IRB.
- And, a lot of other responsibilities – check out the HRPP Plan for the complete listing.
Things happen. There may be questions, concerns, complaints, allegations of undue influence, allegations or findings of non-compliance, or input regarding the HRPP.
If one of these things happen, what do you do?
If an event occurs that is related to a research study, we call this a “Unanticipated Problem Involving Risks to Subjects or Others” or “UPIRTSO”.
So, why is an UPIRTSO important?
As the research has not yet taken place, when an IRB approves or provides a determination for a study, it is a “predictive” approval or determination based on the information provided in the IRB submission.
During the conduct of the study, it is the responsibility of the study team to let the IRB know when something unexpected happens. By doing so, the study team keeps the IRB apprised on whether a reevaluation of risks is needed. This is where the reporting of an UPIRTSO comes into play.
So, what events are considered an UPIRTSO and what needs to be reported?
Any incident, experience, or outcome that meets all of the following criteria must be reported to the IRB:
- Is unexpected, serious, and would have implications for the conduct of the study, or is unexpected (in terms of nature, severity, or frequency) given 1) the research procedures that are described in the protocol-related documents, such as the IRB-approved research protocol and informed consent document; and 2) the characteristics of the subject population being studied;
- Related or possibly related to a subject’s participation in the research; and
- Suggests that the research places subjects or others at a greater risk of harm (including physical, psychological, economic, or social harm) related to the research than was previously known or recognized.
So, let’s say an UPIRTSO happened, how soon do you have to report it?
Harvard University Area IRB policy states that an UPIRTSO needs to be reported to the IRB within five business days; of course, the sooner the better.
Now that we know what an UPIRTSO is and when it has to be reported, how does one report it?
A Report of New Information (RNI) is the method by which to do so. Instructions on how to complete a RNI may be found here.
Interested in more information? Check out these resources:
- HUA Investigator Manual – found in the ESTR Library under the “General” tab. Go to the section on “Prompt Reporting Requirements”.
- HRP-024 – New Information - found in the ESTR Library under the tab “Standard Operating Procedures”
What if something happens and it is not a UPIRTSO or if you have concerns or a question?
Any of these may be reported to the IRB Chair, the IRB Office, the IO/OO, to Legal Counsel (Harvard Office of the General Counsel), Deans, or Department Chairs. Know that Harvard employees are permitted to report concerns on an anonymous basis. Also, those that share a concern, have a question, or anything else that is reported should not (and will not) be subjected to retaliation or harassment as a result of reporting. Concerns about possible retaliation should be immediately reported to the IO/OO or designee.

